- | Healthcare Healthcare
- | Policy Briefs Policy Briefs
- |
Creating Strategic State Stockpiles to Institutionalize Pandemic Preparedness
How states institutionalize pandemic preparedness following the COVID-19 pandemic will determine their ability to cope with the next public health crisis. States with major metropolitan areas should consider building independent, state-specific emergency medical stockpiles to lessen their dependence on the Strategic National Stockpile (SNS) and to be better prepared to manage shortages of critical emergency medical equipment.
Emergency Preparedness Is Both a Federal and a State Concern
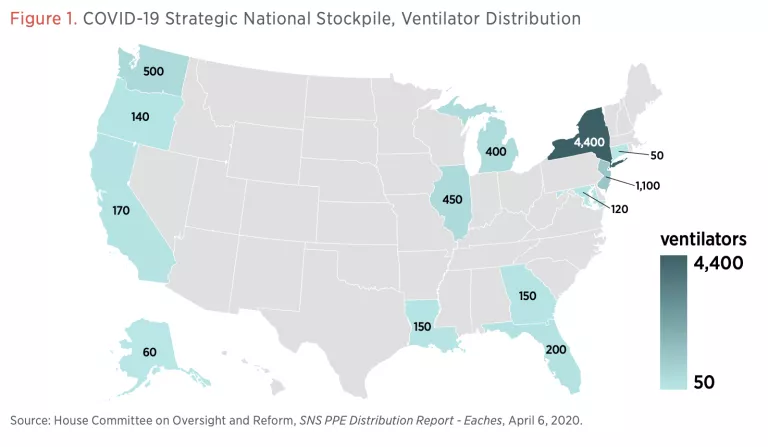
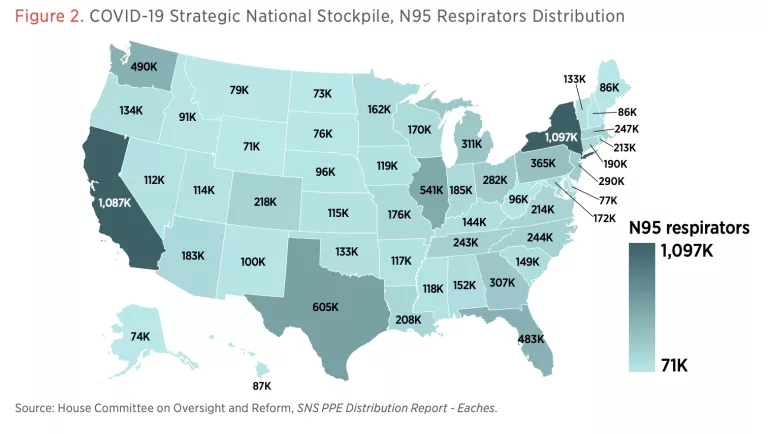
The initial wave of COVID-19 exposed the shortfalls of the SNS, with ventilators and personal protective equipment (PPE) (i.e., N95 masks, surgical masks, gloves, face shields) lacking for patients and healthcare workers (see figures 1 and 2). The failures of the SNS to provide enough emergency medical equipment during the pandemic highlight the importance of creating strategic state stockpiles and more generally institutionalizing pandemic preparedness at the state level.
Large states, especially those with major metropolitan areas heavily affected by COVID-19, should aggregate preexisting medical emergency stockpiles and allocate yearly investments toward developing strategic state stockpiles. These stockpiles will provide two important benefits by (1) avoiding dependence on supplies from the SNS and (2) allowing for faster localized responses to heavily hit cities during pandemics. Specifically, states such as New York, which experienced shortages in ventilators and PPE during COVID-19’s initial wave, should immediately begin stockpiling statewide resources in anticipation of a possible fall or winter COVID-19 return concurrent with seasonal influenza. While not every state may need to create a strategic state stockpile, for the few that do, it should be an essential part of disaster preparation.
Several States Urgently Need a Strategic Stockpile
While the economic consequences of COVID-19 are being felt nationwide, the public health effects of the virus remain relatively regionalized. To date, densely populated urban areas have borne the brunt of the virus’s opportunistic nature; approximately one-fifth of the country’s confirmed COVID-19 cases are in New York City. Other major cities, such as Baltimore, Boston, Chicago, Detroit, Los Angeles, and New Orleans, were also heavily affected. Rural states, without major metropolitan areas, were fortunate to be insulated from the worst of the COVID-19 pandemic. Notably, the five states that never issued shelter-in-place or “safer at home” directives (Arkansas, Iowa, Nebraska, North Dakota, and South Dakota) were predominantly rural in composition. The effect of COVID-19 on these five states was markedly different from that on states with major metropolitan areas (e.g., California, Florida, Illinois, Louisiana, Massachusetts, Michigan, New York, and Pennsylvania).
Even within individual states, the risks of COVID-19 are not evenly distributed. On April 4th, hospitals around New York City prepared for an unprecedented surge in demand. Yet most counties in the state reported fewer than 50 cases, with only Albany, Erie, and Monroe counties confirming more than 300 infections outside the New York City area. Owing to the disparate impact viruses can have on different communities, even within the same state, a state-based stockpile could provide adequate supplies to an affected region effectively and expeditiously in the time it would take for the federal government to mobilize.
The Strategic National Stockpile Was Built as a Temporary, Not a Long-Term, Solution
The initial iteration of the SNS began as the National Pharmaceutical Stockpile Program in 1999, under the auspices of the Clinton administration. It sought to guard against a potential threat of a bioterror attack in a major metropolitan area. The George W. Bush administration renamed the program and expanded the SNS’s capacities in the aftermath of the 9/11 anthrax attacks, SARS, and Hurricane Katrina to encompass emergency medical supplies beyond medical countermeasure pharmaceuticals. The SNS continued to grow under the Obama and Trump administrations, commanding $705 million in annual investments and housing $8 billion in supplies, ranging from smallpox vaccines to ventilators.
Federal leaders never intended for the SNS to provide emergency medical equipment to every state in the event of a prolonged, nationwide pandemic. Instead, the SNS was designed to serve as a resource for acute, regionalized public health emergencies and as a stopgap between the time existing supplies are exhausted and the time the private sector ramps up production to meet the new demand. Moreover, America’s healthcare system operates on an assumption that unaffected states can help heavily affected states during an emergency, which is an effective strategy during hurricanes or wildfires, but not as much during pandemics. For instance, in mid-March the SNS contained 30 million surgical masks and 12 million higher-filtration N95 masks; by early April, nearly 90 percent of the SNS’s total PPE became depleted. The remaining 10 percent of SNS supplies were kept in reserve for a worst-case public health emergency scenario.
States Should Prepare as If the Best-Case Scenario for COVID-19 Treatment Will Not Be Realized
The threat of COVID-19 may not resolve in short order. Antiviral treatments or a COVID-19 vaccine could take longer to develop than is anticipated in the best-case scenarios. According to the Milken Institute’s COVID-19 Treatment and Vaccine Tracker, over 150 treatments for COVID-19 are in preclinical or clinical trials; admittedly, there is great reason to hope that one or several of these trials will yield an effective therapeutic or vaccine. Indeed, clinical trials showed that Gilead’s antiviral therapeutic, remdesivir, accelerated recovery in advanced COVID-19 patients. However, as promising as this is, an effective antiviral therapeutic is not the same as a vaccine.
Historically, antiviral medicines proved more difficult to develop than antibacterial therapies (i.e., antibiotics), mostly owing to antiviral medicines’ inability to destroy the target pathogen, since viruses integrate into a host’s genome surreptitiously and utilize the host’s cellular machinery to replicate. Thus, most antiviral drugs inhibit viral development without damaging the host, and they typically require years of research. Moreover, multiple attempts to create a successful coronavirus vaccine have never proved successful, including attempts for SARS-CoV-2 predecessors MERS-CoV (2012) and SARS-CoV-1 (2003). While the biopharmaceutical industry is making extraordinary efforts to develop a SARS-CoV-2 vaccine—such as the US Department of Health and Human Services’ $483 million investment in Moderna—these extraordinary efforts are no guarantee of success. It is simply not an easy task to develop a vaccine even with the urgency of global pandemic, as is shown by the cases of HIV and hepatitis C.
Owing to this uncertain timeline, each state’s emergency preparedness will be critical in meeting the potential second COVID-19 surge in conjunction with seasonal influenza. The stockpiling and distribution of ventilators, N95 respirator masks, and other PPE will be critical in attending to potential public health emergencies. Creating state strategic stockpiles will allow states to be prepared for a second wave of COVID-19 infections and set the foundation for preparations for future pandemics and emergencies.
Recognizing Pandemic Preparedness as a Long-Term Investment
Institutionalizing pandemic preparedness funding will require calculated decisions by states within their yearly budgets. Admittedly, advocating for increased statewide investments in healthcare may prove difficult, since education and health services already take up substantial portions of state budgets. For instance, Ohio’s fiscal year (FY) 2020 budget allocated 36.78 percent to Medicaid, 8.04 percent to health and human services, 15 percent to primary and secondary education, and 3.54 percent to higher education, for a collective 63.36 percent of the state budget. Similarly, New York’s FY 2021 budget apportioned 22 percent to Medicaid, 27 percent to school aid, and 7 percent to universities. Likewise, Texas’s FY 2018–2019 budget of $179.4 billion allocated $62.4 billion to Medicaid, $4 billion to behavioral health, $59.9 billion to public education, and $20.5 billion to higher education, all of which add up to nearly 82 percent of the state budget.
Parallels between Pandemic Preparedness and Higher Education Investments
The case for supporting pandemic preparedness funding is analogous to that of investing in higher education. The return on investment for pandemic preparedness takes time to materialize. However, when those benefits do materialize, they are invaluable. For instance, Jonas Salk’s 1955 discovery of the polio vaccine at the University of Pittsburgh provided an incalculable benefit to society after nearly a decade of research. Moreover, University of North Carolina researchers are developing the most effective antiviral to date, Gilead’s remdesivir. Likewise, Johns Hopkins University researchers are developing in-home COVID-19 screening tests to allow health systems to test up to 1,000 people per day. Just as investments in higher education can prove to be invaluable in the long term, so too can investments in pandemic preparedness.
The societal and economic response to the COVID-19 pandemic may have been necessary and justified, but it heavily taxed society. Given an estimated real unemployment rate of 20.6 percent and the potential for national unemployment claims to reach 41 million this summer, the COVID-19 pandemic has clearly disrupted all facets of American life. If states remain unprepared, the economic distress of a second wave in the fall, concurrent with the seasonal flu, could be devastating. While healthcare and education represent the majority of state budgets, strategic investments in pandemic preparedness are urgent.
Reducing Economic Uncertainty and Instilling Public Trust
For state economies to cope with the crisis and initiate a recovery, the public needs to regain confidence in the government. That confidence is directly related to signals of policy stability. In particular, the public health response to the pandemic must be (and must be perceived as) planned and consistent, rather than improvised and shifting. It is worthwhile recalling that high policy uncertainty drives down private investment. Consequently, a way to signal a steady hand at the helm is to institutionalize of preparedness, and few plans can have such a quick and tangible instantiation as building a strategic state stockpile.
The Trump administration’s lack of transparency regarding the allocation of the SNS’s supply of ventilators left governors exercising unconventional measures, such as pleading on Twitter or cable television, to obtain them. For instance, Governor Jared Polis (D-CO) made an official request for ventilators via the Federal Emergency Management System and appealed to the vice president, yielding zero ventilators. However, when Senator Cory Gardner (R-CO) contacted the administration directly, the state received 100 ventilators the following day. Similarly, Governors J. B. Pritzker (D-IL), Gavin Newsom (D-CA), and Laura Kelly (D-KS) experienced difficulty receiving ventilators from the federal government. Invariably, the lack of transparency in the application for, approval of, and distribution of emergency medical resources was not a template for optimal public policy—before, during, or after a pandemic.
States can be more transparent about the application and allocation of state strategic stockpile resources because, in contrast to the federal stockpile, each state has only one state-claimant to those resources. All stakeholders—hospitals, nursing homes, grocery stores, schools, small businesses, and citizens—need to know that the process of applying for and receiving materials is both fair and transparent.
States do not possess the same buying power as the federal government, but they possess more precise knowledge of the needs of the state and how to strategically apportion limited state funds. For instance, state strategic stockpiles should focus on bulk-purchasing less expensive but highly essential PPE to supply the healthcare workforce, essential workers, and (potentially) the general population. More expensive investments, such as the stockpiling of respirators, should be made only strategically to maximize available, but limited, state funds.
Moreover, mass supplies of preexisting PPE will allay the high prices and demand seen during the height of the pandemic. In April, FEMA awarded a $55 million contract to a small, private company for N95 respirator masks. FEMA paid the contractor $5.50 per mask, while the federal government typically pays $0.63 per mask with its chosen manufacturer, 3M—representing a 773 percent difference in pricing. Ironically, $5.50 per mask is far higher than what a private citizen might have paid to purchase N95 respirator masks from The Home Depot in May 2020. As of May, private citizens could purchase a 15-pack of N95 masks for $34.97, or $2.33 per mask—half the rate the federal government paid a month ago.
Similarly, ventilator prices have soared during the pandemic. In September 2019, the federal government ordered 10,000 Trilogy Evo Universal ventilators from the Dutch company Royal Philips N.V. at $3,280 per unit to fortify the SNS. However, no units made it into the national stockpile. Nevertheless, commercial versions of the product were available for purchase during the pandemic at $17,154 per device, with state governments and hospitals looking to purchase during the height of the pandemic. Thus, state strategic stockpiles should invest in strategic quantities of low-cost ventilators and concentrate investments in larger quantities of PPE (in particular, N95 masks for frontline healthcare workers and essential businesses).
From Idea to Implementation
During the initial stages of the COVID-19 pandemic, the public looked to federal and academic authorities for guidance on areas outside their expertise. Medical experts on the Coronavirus Task Force, such as Deborah Birx and Anthony Fauci, have provided credibility and reassurance in the federal government’s efforts to mitigate the pandemic. Ultimately, the “hard pause” on American life helped “flatten the curve” and prevented tens of thousands of unnecessary deaths stemming from an overwhelmed medical system. However, as America transitions from a “hard pause” to a “soft restart,” states will take center stage in reopening the economy, and a consortium of diverse statewide voices should be involved in the decision-making processes for future strategic state stockpiles.
States Should Establish an Independent, Nonpartisan Advisory Committee Comprising Public Health Experts, Healthcare Workers, Economists, Small Business Owners, Public Educators, and Citizens
In April, Governor Mike DeWine (R-OH) noted that “none of us is as smart as all of us.” Governor DeWine’s philosophy is an instructive call for democratic decision-making and diverse stakeholder engagement, especially when designing a diverse independent advisory committee for pandemic preparedness that, inter alia, recommends how to create and supply a strategic state stockpile. For the economy and medical system to remain stable in the event of another prolonged pandemic, diverse perspectives must inform on the emergency medical supply needs of various sectors across the state.
Frontline healthcare workers can inform the commission of the daily, weekly, and monthly needs for specific types of PPE when treating patients in hospitals and clinics (e.g., face shields, N95 respirator masks, surgical masks, surgical gloves). Likewise, public health experts from academic and nonacademic institutions (healthcare nonprofits) can provide big-picture epidemiological perspectives and infection estimates to compare to existing strategic state stockpile levels. From an alternative viewpoint, economists can provide insight into the financial tradeoffs of partially or fully closing the economy, identifying which civilian businesses may be most essential for the economy, and thus prioritizing PPE distribution. Similarly, small business owners can discuss what their needs are to remain open, or reopen, with proper amounts of face masks, gloves, and hand sanitizer for employees to feel safe. Alternatively, public schoolteachers can provide insights into the PPE and sanitation equipment they will need for in-class instruction. Furthermore, citizens can provide insights into their PPE needs to resume activities like shopping, attending church services, and other needs of everyday life. Communities of color, which have been disproportionally affected by COVID-19, should have a prominent voice in this commission. Collective insights from these stakeholders can inform leadership about the appropriate distribution of strategic state stockpile resources.
Prioritize Supplying PPE and Ventilators to Nursing Homes and Residential Care Facilities First, Then to Urban Hospitals
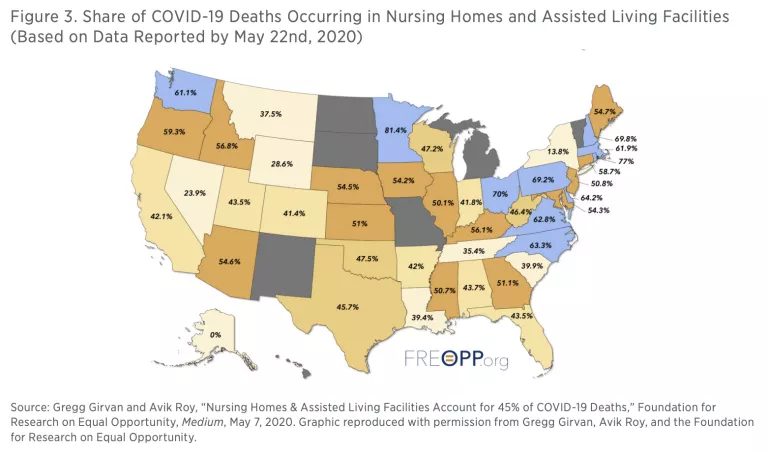
COVID-19 has not affected all age demographics equally. The 0.6 percent of the population living in nursing homes or residential care facilities accounts for 45 percent of all US COVID-19 deaths (see figure 3). Tragically, international data from Australia, Belgium, Canada, Denmark, France, Germany, Hong Kong, Hungary, Ireland, Israel, Norway, Portugal, Singapore, and Sweden show that similar figures of COVID-19 fatalities have occurred at nursing home facilities. Moreover, the 2017–2018 seasonal influenza data from the Centers for Disease Control and Prevention (CDC) showed that of the 61,099 Americans who died, 50,903 of them, or 83 percent, were 65 or older. If a second wave of COVID-19 occurs concurrent with the seasonal flu, the impact on nursing home and residential care facilities could be devastating. To preempt this human tragedy, these communities should be prioritized for PPE distribution from strategic state stockpiles. Protecting these vulnerable populations along with the healthcare workers tending to them will be paramount in protecting the most vulnerable patient populations.
Once Nursing Homes and Healthcare Workers Are Adequately Outfitted with PPE, Supply Essential Workers with Masks
In the event that SNS supplies and strategic state stockpile supplies are able to deliver adequate PPE to nursing homes, hospital systems, and healthcare providers during a pandemic, additional available units should be distributed to essential workers and the public, as per the recommendations of the independent advisory council. New Hampshire serves as a salient case study.
To date, New Hampshire has experienced relatively few COVID-19 cases, with 3,721 testing positive for the disease and 182 deaths. These figures allowed the state to begin an online portal for businesses to complete a mask order form to outfit employees with face masks for months. Within this framework, businesses with essential workers (e.g., grocery store cashiers, CVS and Walgreens associates, and Amazon and UPS delivery personnel) applied for mask allocations and the masks were picked up at 14 different locations throughout the state by business owners. This is a unique model for states to follow, especially in protecting essential workers and limiting the spread of infection between individuals. Ultimately, the benefits of public face coverings are twofold: to protect vulnerable populations from asymptomatic carriers (more so than providing an impenetrable barrier for the user) and to prevent fomite transmission by touching one’s nose or mouth.
Providing PPE to essential workers has also proved successful in heavily affected New York City, as law enforcement officers, healthcare workers, and transit operators have reported lower coronavirus infection rates than the general population. Nevertheless, states will only be able to provide PPE to essential workers if enough supply exists to outfit the healthcare providers on the front lines. The confluence of state strategic stockpile supplies and SNS provisions will provide the resources necessary to transition this idea into practice.
Conclusion
Pandemic preparedness is often thought of as an abstract concept. However, stockpiles make the abstract concrete and will provide a material foundation to build upon to mitigate the effects of a future pandemic. What’s more, strategic state stockpiles can be the catalyst of the institutionalization of pandemic preparedness and, equally importantly, signal to citizens and businesses that state governments will no longer improvise, but will respond to the next wave and the next pandemic in a planned and consistent manner.
About the Author
Mark E. Dornauer is a visiting fellow at the Foundation for Research on Equal Opportunity. Previously, he worked at the US Department of Health and Human Services Office of the Assistant Secretary for Preparedness and Response, where he edited public health emergency preparedness and pandemic response strategies. There, he oversaw the writing of strategies relating to the Strategic National Stockpile (e.g., ventilators, PPE) and medical countermeasures (e.g., vaccines). He was educated at the University of Pennsylvania, Boston College, and Georgetown University.

