- | Healthcare Healthcare
- | Policy Briefs Policy Briefs
- |
A Snapshot of Healthcare Regulations in Southeastern States
Healthcare in the United States is complex and expensive, a consequence of decades of regulatory accumulation. The relationship between federal and state regulations has further complicated the effects of regulation on the healthcare industry. Certificate-of-need (CON) laws, occupational licensing laws, scope-of-practice laws, reimbursement regulations, insurance coverage regulations, and myriad other regulations have resulted in one of the most expensive healthcare systems in the world, and they have resulted in outcomes that are directly are odds with the stated purpose of the regulations. For example, CON laws, which regulate the major capital expenditures of new and existing hospitals, have led to 99 fewer hospital beds per 100,000 people. Occupational licensing restrictions have discouraged people from changing their job or moving to another state. Although not all regulations are necessarily detrimental, their sheer volume means that they are likely to result in some adverse outcomes.
All around the nation, spurred by the COVID-19 pandemic, states have been reviewing how to use existing regulations to address challenges in healthcare delivery. Telehealth, occupational licensing, scope of practice, and CON have all received the attention of scholars and policymakers as tools for responding to the pandemic and for improving health outcomes in the long term.
This snapshot is the first in a series that will compare the healthcare regulatory landscapes and highlight areas for reform across states, beginning with the Southeast region of the United States. The Southeast region, as defined by the Bureau of Economic Analysis, comprises Alabama, Arkansas, Florida, Georgia, Kentucky, Louisiana, Mississippi, North Carolina, South Carolina, Tennessee, Virginia, West Virginia. We use public data and proprietary data produced through the Quantitative Health Lab (QHL), an initiative of the Open Health Program at the Mercatus Center at George Mason University.
The rest of this snapshot proceeds as follows: We first briefly describe our primary data sources. Then we summarize healthcare regulatory restrictions in the Southeast region, compare the volume of healthcare regulatory restrictions to that of other types of regulation, and consider the volume of each state’s regulatory restrictions in the context of each state’s size. We show that healthcare regulations represent a significant portion of state regulations. After that, we discuss specific types of regulations, including CON laws, scope-of-practice laws, telehealth regulations, and occupational licensing restrictions in healthcare. Next, we review health outcomes and health services in the region, and we also discuss the Healthcare Openness and Access Project (HOAP) index scores for the region. Following that, we briefly discuss state-level health outcomes in the region, and we conclude the snapshot by highlighting some opportunities for reform.
DATA SOURCES AND METHODOLOGY
We use data from multiple sources, including unique data generated through the QHL. These data come from the RegData suite of datasets, namely State RegData, State Healthcare RegData, and Occupational Licensing RegData. In addition, we include other data compiled by Mercatus scholars such as data on the prevalence of CON laws, HOAP index scores, and data on scope-ofpractice regulations. Each data source is described in the appendix, with a particular emphasis on RegData.
STATE HEALTHCARE REGULATORY RESTRICTIONS
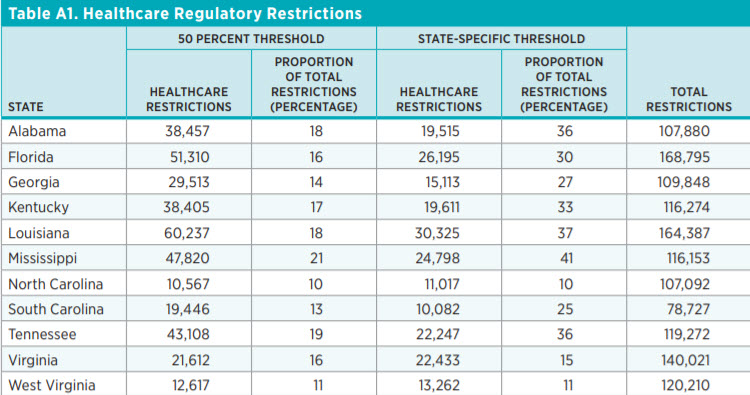
The simplest way to quantify healthcare regulations is to count the number of regulatory restrictions. State Healthcare RegData contains these counts. Figure 1 shows the variations in regulatory restrictions across the Southeast region. The light teal bars show the total number of regulatory restrictions in each state, and the dark teal bars show the total number of healthcare regulatory restrictions.
In terms of total regulatory restrictions, Florida (168,796) leads the region, with Louisiana (164,387) in a close second place, and South Carolina (78,727) has the fewest restrictions. However, in terms of healthcare restrictions, Louisiana (60,237) leads the region.
Figure 2 shows healthcare regulatory restrictions as a share of total regulatory restrictions across the region. There is no direct correlation between the total number of restrictions and the number of healthcare restrictions. The number of restrictions can vary depending on how important an industry is to a state. For example, in West Virginia, the mining sector is the third-greatest contributor to state GDP, and among all the states in the region, West Virginia has the greatest number of restrictions on mining.
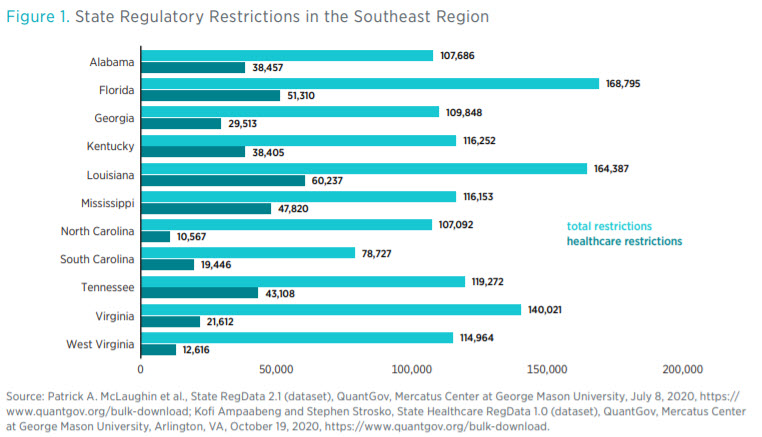

In the Southeast region, South Carolina and North Carolina are two of the least regulated states in that they have the smallest number of total restrictions and the smallest number of healthcare restrictions, respectively. The proportion of healthcare restrictions to total restrictions in North Carolina is also the smallest in the region. Furthermore, North Carolina has the fewest healthcare restrictions per capita (as shown in figure 3). Louisiana has the greatest number of healthcare restrictions, but Mississippi’s healthcare restrictions make up the largest share of total regulatory restrictions in the region.
HEALTHCARE RESTRICTIONS PER CAPITA
Often regulations are promulgated to address specific local concerns. Other researchers have noted that population density is a good predictor of the volume of regulations in a jurisdiction. Taking this fact into consideration, figure 3 shows the number of healthcare regulatory restrictions per capita. For readability, the numbers in figure 3 are inverted—that is, they are the number of residents in the state that are served by a single regulatory restriction. Using this metric, Florida, the state with the second-highest number of healthcare regulatory restrictions, is actually the sixth among the Southeast states when adjusted for population. These numbers are consistent with previous research showing that states with greater populations are more regulated.
HEALTHCARE RESTRICTIONS AND INDUSTRY CONTRIBUTION TO STATE GDP
One of the key advantages of using State Healthcare RegData is that we can determine the industries or sectors of the economy that are likely to be affected by regulation. Thus, we can determine industry-relevant regulatory restrictions. For the purpose of this determination, we define industries according to the North American Industry Classification System. In this section, we examine the relationship between the size of healthcare-related industries in each state and the number of regulatory restrictions each state places on those industries.
Figure 4 shows the top five industries in each of the Southeast states as measured by the contribution of the industry to state GDP. Healthcare-related industries include ambulatory healthcare services, hospitals, and nursing and residential care facilities. In 9 of the 11 Southeast states, either ambulatory healthcare services or hospitals is among the top five industries. Though not causative, the size of an industry in a state does influence the number of state regulatory restrictions thereon. For example, in Florida, the state with the second-most healthcare regulatory restrictions, ambulatory healthcare services are the second-greatest contributor to GDP. The relationship is not perfect, though. Louisiana has the most regulatory restrictions on healthcare-related industries, but the ambulatory healthcare services industry ranks fifth in state GDP contribution. This circumstance warrants a deeper look.
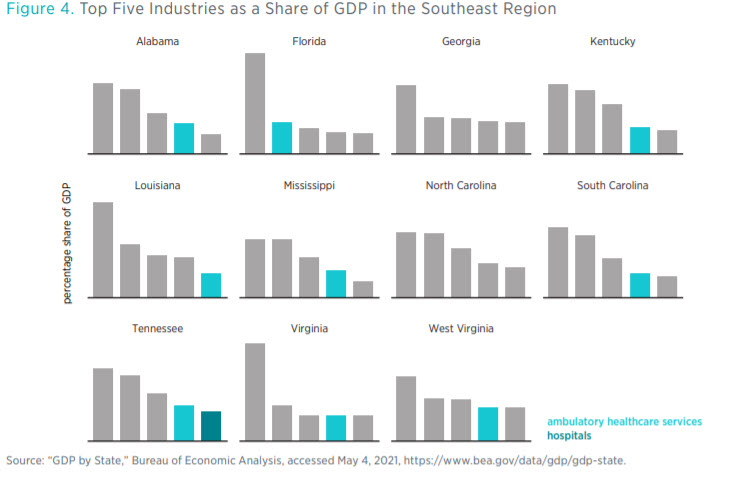
Florida’s case may be reflective of the demographics of the state, given that 20 percent of the state’s population is 65 or older and requires greater levels of spending on geriatric healthcare.
These simple but innovative ways of measurement provide an overview of the pervasiveness of healthcare restrictions within a state but do not measure the effect those restrictions have on those being regulated—providers and patients. Measuring this effect requires a more in-depth look at particular healthcare occupations and at other regulations that affect the delivery of healthcare.
SPECIFIC HEALTHCARE REGULATIONS
In the previous section we summarize the volume of healthcare regulatory restrictions in the Southeast region. However, healthcare is a vast field and there are different types of regulations such as CON laws; occupational licensing laws; scope-of-practice laws; and regulations on healthcare facilities, insurance, and drug prices. In this section we introduce in more detail each type of regulation and compare states in the Southeast in terms of the volume of such regulations.
Given the wide breadth of healthcare regulations, we leave many topics unexplored, such as state prescription drugs price controls. In addition, our review of scope-of-practice laws does not include nurse practitioners and examines only one aspect of pharmacy practice. This speaks to the breadth of healthcare regulations in the states.
Certificate-of-Need Laws
CON laws require medical providers to seek government permission before embarking on major capital expenditures such as building new hospitals, installing new equipment, providing new services, or expanding existing buildings. Although legislators have enacted CON laws to increase healthcare quality and supply, research shows that the laws tend to achieve the opposite.
Mercatus scholars and other academic researchers have established that CON laws produce no discernible benefits other than protection for incumbent healthcare providers. They lead to shortages of hospital beds and medical imaging equipment and have a negligible or negative effect on health outcomes.8 The Mercatus Center’s CON laws database and research show that repealing CON laws would have significant positive effects on healthcare delivery and health outcomes.
The Southeast region is unique in that it is the only region where every state has a CON law on the books. North Carolina, with 27 CON laws (see figure 5), has the highest number of CON laws in the region. Actions for which every state in the region requires a CON include
• buying nursing home beds or long-term care beds,
• building or expanding intermediate care facilities for individuals with intellectual disabilities,

• offering psychiatric services,
• offering substance and drug abuse services (except Louisiana), and
• building new hospitals or making hospital-sized investments (except Louisiana).
Reform or repeal of these regulations has been the trend for the past 40 years. Currently, nationwide, 11 states do not have any CON laws, and 4 have only one CON law. The Southeast region has seen some reforms in states such as Florida, but it still lags the rest of the country.
Regulation of Medical Professionals
Healthcare occupations can be regulated in multiple ways that restrict where healthcare professionals can practice, how often they can practice, and what tasks they can perform. Occupational licensing laws and scope-of-practice laws are the two main sources of regulation in this area. Occupational licensing laws determine who can work in certain occupations, and many healthcare occupations require a license. Scope-of-practice determines how broadly healthcare professionals can employ their training. For example, such laws may require physician assistants and nurse practitioners to practice only with supervising physicians.
The number of regulatory restrictions on an occupation may relate to the number of individuals employed in that occupation. Figure 6 shows the number of physicians and surgeons per 100,000 people in each state. The state with the greatest number, Tennessee, has almost 100 physicians and surgeons per 100,000 people more than the state with the smallest number, Mississippi. This disparity
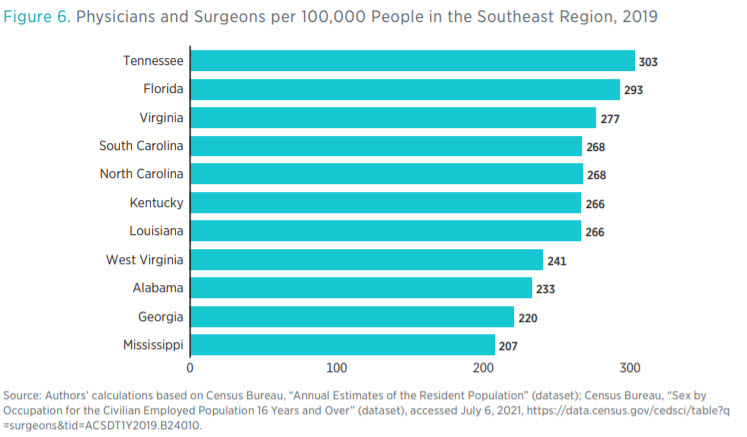
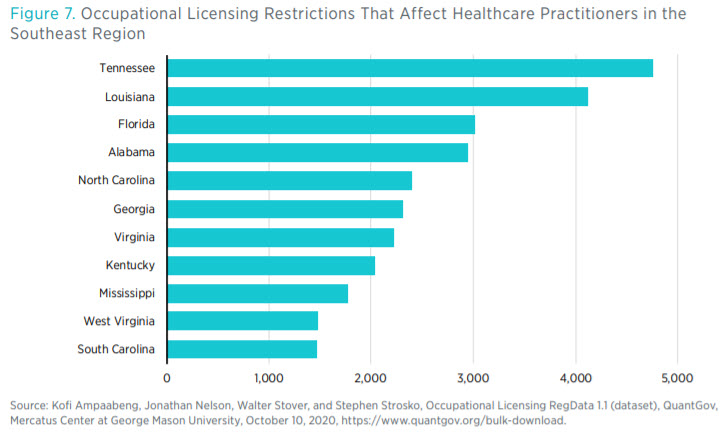
closely matches the disparity between the number of regulatory restrictions over certain healthcare occupations (see figure 7). Having said that, we do not claim any direct causal connection between the volume of regulations and the supply of healthcare inputs, such as the number of physicians.
Occupational Licensing RegData allows one to look at how restrictions may affect different occupations (as defined by the Standard Occupational Classification system). Figure 7 shows the number of regulatory restrictions that affect healthcare practitioners.11 There is a noticeable difference between the number of restrictions on healthcare practitioners and the number of restrictions on healthcare support occupations.
Multistate Practice and Compact Participation
Licensure of medical professionals is state based, which means that providers must obtain a license from each state in which they wish to practice. Although medical education standards are practically uniform across the country, each state imposes its own licensing requirements. This arrangement is costly and time consuming for practitioners who want to practice in multiple states or even move to another state to practice. The result of state-based licensing is that medically qualified professionals are less likely to move between states. A growing body of research shows that licensed workers are less likely to move to other states than their unlicensed counterparts.12 As was evidenced during the COVID-19 pandemic, mobility of medical professionals is key to improving access to healthcare.
To increase the geographic mobility of medical professionals, various professional organizations have proposed legislation to create compacts among states that streamline the licensing process. Though these compacts come in different forms, they commonly allow licensed professionals to practice in all participating states. Members of different medical professions have created or proposed seven interstate compacts: the Interstate Medical Licensure Compact (for physicians); the enhanced Nurse Licensure Compact (eNLC); the Advanced Practice Registered Nurse Compact; the Emergency Medical Services Compact; the Audiology and Speech-Language Pathology Interstate Compact, the Physical Therapy Compact, and the Psychology Interjurisdictional Compact. In order for a compact to take effect, the state legislature must adopt model legislation that contains the relevant provisions of the compact and the condition(s) by which the compact takes effect.
Most states have signed onto such healthcare compacts, promising to respect out-of-state medical licenses within their borders. The NLC, which was the first of the compacts and the most widely adopted (with 35 participating states), allows board-certified nurses to practice in any participating state without obtaining a new license. By participating in compacts, states benefit from a national pool of providers, especially during emergencies and other disasters. Licensure compacts also facilitate telehealth, wherein physicians within a member state can practice virtually with patients in other participating states.
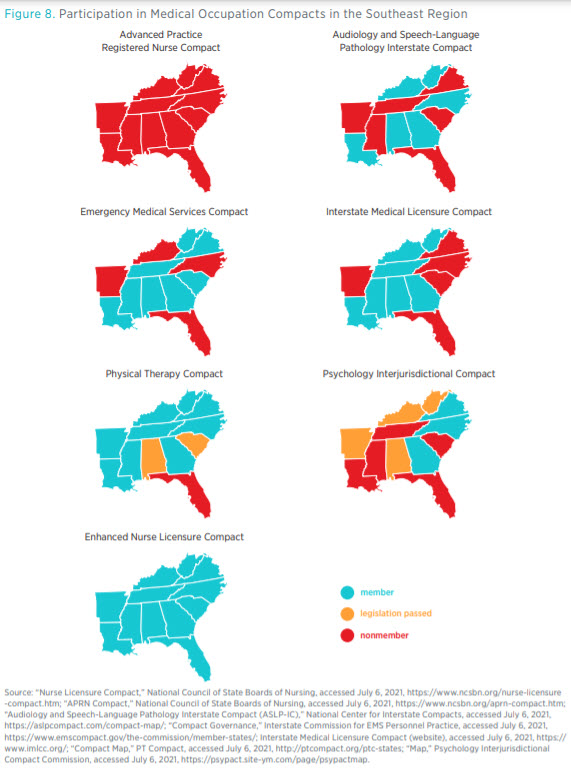
Figure 8 shows the membership of medical professional compacts across the Southeast. As of June 2021, all states in the region were members of the eNLC. Georgia, Mississippi, North Carolina, Tennessee, and West Virginia were members of four or more compacts each, whereas Florida was a member to only one compact, the eNLC. No state in the region has yet joined the Advanced Practice Registered Nurse Compact, which was created in 2020 and presently has only North Dakota as a member. In addition, a number of states have enacted the necessary laws to join existing compacts.
Telehealth Regulations
Telehealth regulations typically address a number of issues, including coverage, payment, and originating sites. We review the regulatory landscape as it pertains to these three issues. Telehealth coverage regulations typically mandate that health insurance providers or health maintenance organizations cover certain conditions. Payment regulations require, among other things, payment parity, which providers and hospitals routinely demand. Payment parity means that payers reimburse telehealth services and in-person services at the same rate. However, a few states exclude charges such as facility fees from parity rules. Finally, originating site regulations define telehealth in terms of the location of the provider and the patient. Before the COVID-19 pandemic, states, and especially the federal government, reimbursed telehealth services only if the patient was in a rural or underserved area.
Some telehealth regulations not covered in detail in this snapshot include requirements that medical staff be located in or hold a medical license in the same state as their patients when administering telehealth services and state or federal regulations that allow only certain staff to practice telehealth services or limit what services can be provided. For example, according to federal regulation, physicians issuing prescriptions can do so only if the patient has received at least one in-person consultation previously or has another physician present with him or her during a telehealth session. Many of these requirements were temporarily lifted during the COVID-19 pandemic, with some being made permanent through legislative action. A notable example are states signing onto medical licensure compacts, which allow healthcare professionals to practice in all participating states both in person and virtually.
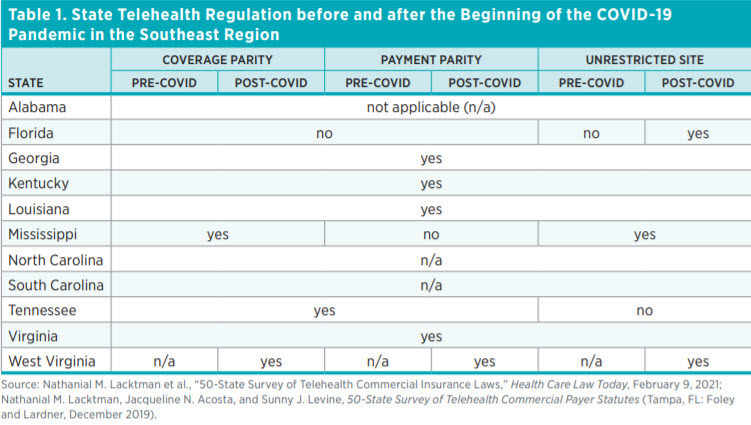
Table 1 shows the state of telehealth laws in the Southeast region along three dimensions: coverage, payment, and originating site. Alabama, North Carolina, and South Carolina do not have any telehealth regulations, whereas Georgia, Kentucky, Louisiana, and Virginia have regulations involving each of the three dimensions. West Virginia has enacted the most changes by mandating coverage and payment parity, and it did not impose restrictions on originating sites. Florida has no coverage or payment parity requirements but has moved to remove limits on originating sites.
The COVID-19 pandemic was a wake-up call for states to reassess their regulatory environments and determine what barriers existed that hindered care. Within the Southeast, West Virginia took the biggest steps in addressing telehealth. Before the COVID-19 pandemic, West Virginia laws did not address telehealth within the three dimensions. New laws passed in 2021 mandate coverage and payment parity. Moreover, West Virginia continues not to impose restrictions on the originating sites for telehealth visits.
Pharmacist Scope-of-Practice Regulations
Several states limit what services nonphysician medical professionals can render. For example, in a number of states, advanced nurse practitioners, who are nurses with advanced degrees, such as a master’s degree or doctorate, are restricted in various ways from practicing to the full extent of their training. Several other professionals are also equally restricted, including pharmacists, physician assistants, and dental hygienists among others. Researchers Gina Oliver and coauthors estimate that restricted scope of practice reduces access to care and causes worse health outcomes. It is instructive that during the COVID-19 pandemic, one of the most effective tools to combat the pandemic has been the temporary relaxation of scope-of-practice restrictions.
We focus here on the differences in vaccination authority granted to pharmacists. In all states, pharmacists, in spite of being highly educated and trained, are unable to administer most vaccinations without explicit authorization from the state legislature. Pharmacists were only recently authorized to administer disease-preventing vaccines. The first training to administer vaccines occurred in 1994, and since then, pharmacists have been instrumental to the increasing uptake of vaccinations.
The authority to administer vaccines is often vaccine specific, may require prescription from a physician, and may even include age restrictions on the patient. Table 2 compares pharmacy vaccination restrictions in the Southeast region. The vaccinations included in the summary are pneumococcal, zoster, Td (tetanus and diphtheria) or Tdap (tetanus, diphtheria, and pertussis), HPV (human papillomavirus), hepatitis B, MMR (measles, mumps, and rubella), and meningococcal vaccines. Alabama, Tennessee, and West Virginia permit licensed pharmacists to administer all seven types of vaccines without any restrictions. In fact, all states in the union, with the exception of New Hampshire, New York, and Wyoming, allow pharmacists to administer pneumococcal and zoster vaccines with full prescriptive authority. (However, these permissions also require that pharmacists be trained.) Georgia requires a prescription for five of the types of vaccines, and North Carolina requires a prescription with one. Six states have an age restriction on vaccines.

HEALTHCARE OPENNESS AND ACCESS INDEX
So far, we have analyzed healthcare regulations in isolation. However, researchers at the Mercatus Center have created an index comprising composite scores of state healthcare regulations. Researchers calculate these scores according to five regulation categories:
1. Professional: How strictly states regulate particular healthcare occupations.
2. Institutional: How strictly states regulate institutions such as hospitals, pharmacies, and insurance companies.
3. Patient: What drugs states allow patients access to and whether there are protections for free speech and Good Samaritans in medicine.
4. Payment: How states restrict or liberate payment arrangements between patients and providers.
5. Delivery: How easy it is for healthcare to be given to patients and to what extent innovation in healthcare delivery is allowed.
Each category itself has a score that is calculated according to certain metrics. For example, professional regulation involves reciprocity of medical licensure, breadth of midwife scope of practice, and the extent of liability of charity caregivers. The purpose of HOAP is to raise important
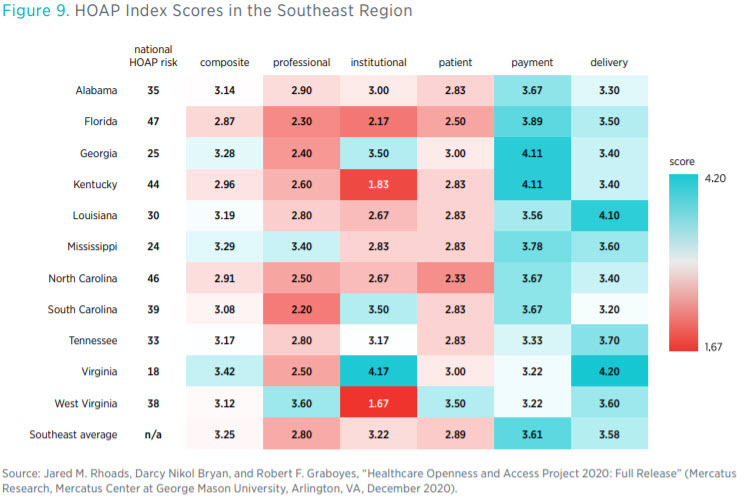
questions, such as how open each state’s laws and regulations are to institutional variation in the delivery of healthcare and how much access to varying modes of care this openness confers on each state’s patients and providers.
Three states in the Southeast (Virginia, Mississippi, and Georgia) have composite scores in the top 25 of the index (see figure 9). Florida, North Carolina, and Kentucky have composite scores in the bottom 10. Scores in each component category paint a more nuanced picture, though. For instance, although Kentucky ranks 44th overall, it ties with Georgia for 1st place in having the best environment to buy healthcare services (by not taxing health savings accounts, among other things).
HEALTH OUTCOMES
Ultimately, the goal of healthcare regulations should be improved healthcare outcomes. As the response to the COVID-19 pandemic has demonstrated, regulatory reforms can improve health outcomes and save lives. Most restrictions we highlight in this snapshot impact the supply side of care—that is, CON laws or occupational licensing laws, for example, determine how much care is provided in a state by restricting the number of physicians or the number of hospitals that can be built. These restrictions can exacerbate poor health outcomes, but greater access to care could help alleviate ambulatory, inpatient, and preventative care gaps. Given the descriptive nature of this snapshot, we do not claim a direct connection between regulatory restrictions and health outcomes. Other studies have examined any such connection. However, by presenting health outcomes in the Southeast region here, we put the regulatory landscape within the appropriate context.
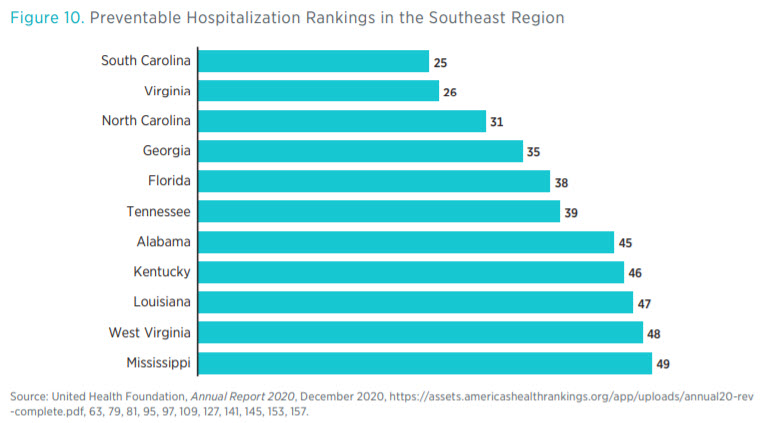
Figure 10 shows a ranking of states in the Southeast region according to their effectiveness in mitigating preventable hospitalizations. Within the region, 5 states rank in the bottom 10 in the nation for preventable hospitalizations.
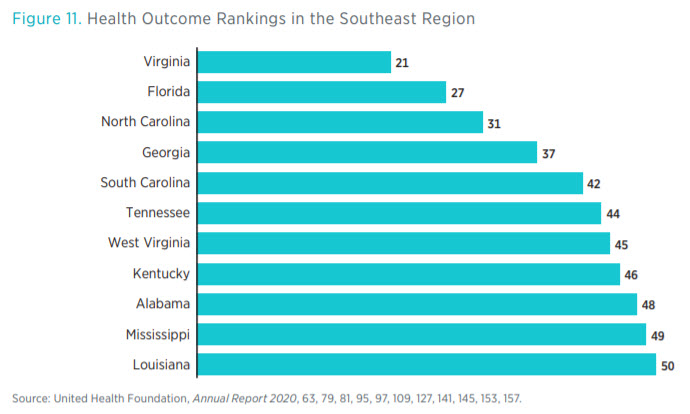
Health outcomes in the region show that the Southeast tends to perform poorly compared to the rest of the country, according to data by the United Health Foundation (see figure 11). Eight states in the Southeast rank in the bottom 10 on overall health outcomes, an aggregate measure of physical health, risk factors, and mortality. Louisiana, Mississippi, and Alabama rank the lowest in the country. Similarly, Mississippi and Louisiana also rank in the bottom 10 in the percentage of patients who seek healthcare but have difficultly finding it (see figure 12).
Looking at these three figures, one can see a negative association between the number of regulatory restrictions and health outcomes. Again, we caution against attributing causality, though some research does examine particular regulations and their effect on health outcomes. For example, research from the Mercatus Center shows that states with CON laws tend to have poorer health outcomes and higher care costs than states without them.

As shown in figure 12, healthcare access may be improved by removing restrictions to support primary care. This can be done by repealing CON laws or telehealth restrictions, which limit the supply of primary care services.
CONCLUSION
In this snapshot of the Southeast region, we present a picture of healthcare regulation by using unique data from the Mercatus Center and other sources. States differ considerably in the volume and scope of healthcare regulations. North Carolina is the least regulated state in the region in terms of healthcare restrictions, whereas Louisiana and Florida are among the most regulated.
Many states still have CON laws, despite substantial evidence that they are detrimental to healthcare access and patient outcomes. At least 11 states in other parts of the nation have repealed CON laws without any adverse effects. If Southeast states wish to improve healthcare access, repealing CON laws is a step in the right direction; these states would also benefit from joining medical professional compacts.
Over the past year, states have made regulatory changes to improve access to care. For example, West Virginia passed laws to facilitate the use of telehealth. Unfortunately, the law also mandates payment parity between in-person and telehealth consultations. Payment parity laws completely ignore the costs of providing services and impose artificially high prices on care, negating an intrinsic benefit of telehealth. Encouragingly, Florida has repealed its telehealth originating site restrictions, allowing for telehealth to be provided regardless of the location of the provider or the patient. Florida also does not mandate telehealth payment parity.
As state governments continue to review their regulations in the wake of the COVID-19 pandemic, policymakers and researchers must have a complete picture of the regulatory landscape. This snapshot is an attempt to provide such a picture. That said, there are other regulations that were necessarily excluded, such as drug price controls and regulations on the scope of practice of other kinds of providers. All these regulations need to be considered for truly comprehensive review and reform to occur.
APPENDIX
Data Sources and Methodology
Measuring Regulations: An Introduction to RegData
State Healthcare RegData
Occupational Licensing RegData