- | Medical Innovation Toolkit Medical Innovation Toolkit
- | Regulation Regulation
- | Data Visualizations Data Visualizations
- |
More FDA Spending Does Not Necessarily Mean Better Results
The FSMA mandates a HACCP-like approach for all food. There is no evidence that this approach is likely to result in any significant progress in lowering the rate of foodborne illness. In view of the experience of the last 20 years, neither increasing the FDA’s resources nor causing the food industry to spend more (in fact, much more) using this approach appears to be the answer.
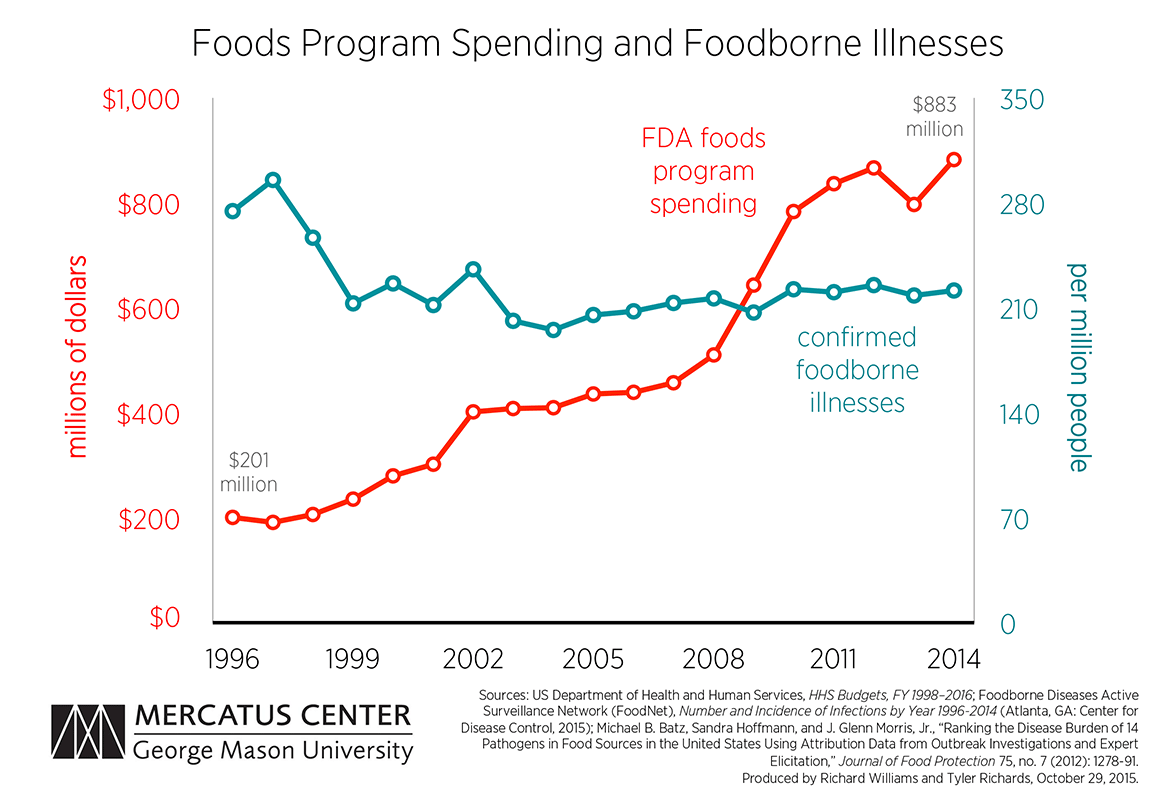
The FDA Foods Program recently requested a $1.167 billion budget for fiscal year 2016. If authorized, this budget would amount to an increase of 27.7 percent ($253 million) above the enacted $914 million for fiscal year 2015. This request is the most recent in a number of steps to implement the Food Safety Modernization Act of 2010 (FSMA), which expanded the FDA’s authority to regulate imported and domestically produced foods. The FSMA is designed to reduce foodborne illnesses in the US, but if recent history is any guide, increasing spending may not be effective.
Using the Foods Program’s yearly budgets, the results of a recent Centers for Disease Control and Prevention report on foodborne illnesses from 1996 to 2014, and a study estimating the proportion of each pathogen-related illness that is attributable to particular food types, we are able to estimate the number of confirmed foodborne illnesses caused by FDA-regulated foods per million people and compare this trend with the FDA’s spending to combat foodborne illnesses. The chart below shows the results of this estimation.
The number of confirmed foodborne illnesses has remained relatively steady for the last 15 years, beginning at 213 illnesses per million people in 1999 and ending at 222 per million in 2014. However, the amount of money spent on the Foods Program has nearly quadrupled over that time.
The only significant drop in foodborne illnesses occurred in the years 1997–1999. This was driven largely by an almost 40 percent decrease in the incidence rate of Campylobacter, one of the most common causes of mild foodborne illnesses. However, it is not obvious this decline was the result of FDA actions, since Food Programs spending saw no unusual increase. Spending went up an average of 10.95 percent over these three years, while the average increase for all 19 years of this study was 9.16 percent.
Although the FSMA is geared toward prevention rather than control, the FDA has had little success with a similar approach in the past. The new FSMA regulations closely resemble a system of controls implemented by the FDA and the USDA called Hazard Analysis and Critical Control Points (HACCP). HACCP began in the 1960s to ensure safe food for space expeditions, but many firms privately adopted it where it was thought to be useful to control pathogens.
In the mid-1990s, the FDA decided to mandate HACCP for some industries. One example of the FDA’s use of HACCP is a seafood rule. The largest expected benefit was a reduction of illnesses caused by the pathogen Vibrio vulnificus. The FDA estimated that the annual number of Vibrio-related cases would decline 20 to 50 percent, and this accounted for half of the rule’s expected benefits. Instead, the annual number of cases nearly doubled over the next 14 years.
Nevertheless, the FDA and the USDA continued with new HACCP regulations.
The FSMA mandates a HACCP-like approach for all food. There is no evidence that this approach is likely to result in any significant progress in lowering the rate of foodborne illness. In view of the experience of the last 20 years, neither increasing the FDA’s resources nor causing the food industry to spend more (in fact, much more) using this approach appears to be the answer.
Notes:
1) Methodology: The Batz, Hoffman, and Morris study estimated the fraction of each pathogen-caused illness that came from particular food groups from 1998 to 2008. Since the USDA also regulates some food products, we reduced the number of foodborne illnesses for each pathogen to match the estimated fraction of illnesses that were caused by FDA-regulated foods.
2) Limitations: This study is limited by the data available on total foodborne illnesses. The figures shown in this chart are estimations based on the best available data from culture-confirmed cases of foodborne illness.