On January 8, 2021, the US Department of Health and Human Services (HHS) announced that it would be finalizing a regulation titled “Securing Updated and Necessary Statutory Evaluations Timely,” which forms the acronym SUNSET. Despite its unassuming name, the regulation is actually one of the more ambitious rule changes to emerge in the four years of the Trump administration.
Regulatory agencies in the federal government are required to conduct periodic reviews of their regulations under Section 610 of the 1980 Regulatory Flexibility Act (RFA). Section 610 requires agencies, consistent with their other statutory objectives, to periodically look back at existing regulations to minimize economic impacts on small businesses. In so doing, agencies are to determine whether rules are still needed, whether they are overly complex, and whether they should be updated to reflect evolving circumstances. The RFA was intended to alleviate regulatory burdens and create regulatory flexibility for small businesses, which are known to be disproportionately burdened by the costs of regulations.
Historically, RFA compliance has been weak. HHS, during the process of promulgating the SUNSET rule, identified just three regulations in the past decade that were finalized in response to Section 610 reviews. HHS estimates that 85 percent of regulations adopted before 1990 had never been edited. As one study recently noted, the periodic review requirement of the RFA has not been complied with consistently “in part because there is no penalty if an agency ignores the RFA.”
HHS’s new SUNSET rule is an attempt to increase compliance with the RFA and to spur more retrospective reviews. It does so by creating a new forcing mechanism according to which, if HHS fails to review a rule in accordance with the RFA, the regulation automatically expires after a predetermined amount of time (in most cases after 10 years). Such an expiration date is known as a sunset provision. The SUNSET rule imposes a sunset provision on the vast majority (with some exceptions) of the approximately 18,000 sections of the US Code of Federal Regulations (CFR) under HHS’s purview.
The RFA review process HHS sets forth in the SUNSET rule works as follows: First, regulations need to be assessed in order to determine if they have a significant economic impact on a substantial number of small entities (SEISNOSE—a term of art from the RFA). If they do, a more in-depth review follows, based on review criteria set forth in the RFA. The first sunset date kicks in five years after the SUNSET rule is finalized, meaning that about 95 percent of HHS regulations will have to be assessed (and potentially reviewed if found to have a SEISNOSE) by the end of 2026 or else the various regulations expire, since most HHS regulations were adopted before 2016 and, thus, will be 10 years old by the time of the first sunset date. Once assessed (and reviewed, if necessary) a regulation’s expiration date is pushed back another 10 years. If a regulation requires updating, HHS has two years to update it, though this deadline may be extended.
HHS’s preamble to the SUNSET rule includes some estimates of the rule’s costs. These estimated costs fall into two categories: (a) costs to the department from allocating personnel to assessing and reviewing department regulations and (b) costs to the public from monitoring and commenting on regulations during the review process. HHS estimates that annualized costs over 10 years would fall in the range of about $8 million to $25 million. However, like most regulatory impact analyses (RIAs) issued by federal agencies, the RIA for the SUNSET rule does not include a monetized estimate of the rule’s benefits.
The purpose of this policy brief is to provide such an estimate of benefits to compare with the department’s estimated costs of the SUNSET rule. This policy brief is organized as follows. The next section provides a benefit estimate for the rule based on some recent HHS experiences with retrospective review. All told, the benefits of the rule are potentially very large, such that it could pass a benefit-cost test, perhaps paying for itself many times over. After that, the brief compares estimated benefits to the costs found in the RIA accompanying the rule, which tend to be about two orders of magnitude lower than the estimated benefits. Later, the brief discusses potential shortcomings of this benefit estimate, which, because it is based on past government RIAs, is subject to considerable uncertainty. The brief concludes that the success of the rule will ultimately depend on which regulations are amended in response to conducting future retrospective reviews. The identities of these regulations are to some extent unknowable. However, the SUNSET rule’s new forcing mechanism gives reason to believe that in the future, retrospective reviews will become a much more prevalent and important part of HHS policy than they have been historically.
Benefits Analysis
Background
The benefits stemming from the SUNSET rule are the net social benefits of whatever regulatory updates, amendments, or rescissions end up occurring as a result of the assessments and reviews that will take place following the rule’s implementation. It is impossible to know with certainty the universe of regulations that will be updated in light of strengthened enforcement of periodic review requirements and better department compliance with the RFA. However, past Section 610 reviews offer some perspective, as does former President Barack Obama’s Executive Order 13563, which ordered a review of existing regulations. Both past experiences can potentially inform an estimate of the benefits of the SUNSET rule.
In order to produce an estimate of benefits, the analysis in this brief makes several assumptions. The first assumption is that the initial assessments (which are conducted to determine whether HHS regulations will have a SEISNOSE) result in no new rulemaking activity on their own. This assumption seems reasonable given that once a regulation is deemed not to have a SEISNOSE, HHS will have fulfilled its retrospective review requirement, and the expiration date will move back 10 years on the relevant regulation. In its final notice of the SUNSET rule, HHS is clear that it contemplates amendment or rescission of regulations that have been reviewed, which is a step that comes after rules are assessed.
The next assumption is that no regulations will accidentally expire owing to HHS not conducting a timely assessment or review. This assumption also seems reasonable, given the experiences of many states with sunset provisions in their laws. For example, in its final notice of the SUNSET rule, HHS points to states such as Idaho, Missouri, New Jersey, and North Carolina, which have sunset provisions for regulations and where accidental expiration of rules seems to be an exceptionally rare phenomenon.
HHS has also built safeguards into the SUNSET rule to prevent inadvertent expiration of regulations. For example, the public will be able to submit comments requesting that HHS commence an assessment or review, and HHS plans to release a list of when all of the regulations under HHS authority were created or last modified, which will allow the monitoring public to determine the expiration date for all or nearly all HHS regulations.
Benefits Assessment
The benefit estimates in this section focus on the reviews expected to be conducted and the corresponding amendments and rescissions that follow from these reviews. As noted, HHS identifies three regulations in the past decade that emanated from its Section 610 reviews. These regulations are presented in table 1, along with the corresponding impacts these regulations were expected to produce, according to the economic analyses accompanying these regulations at the time of their promulgation.
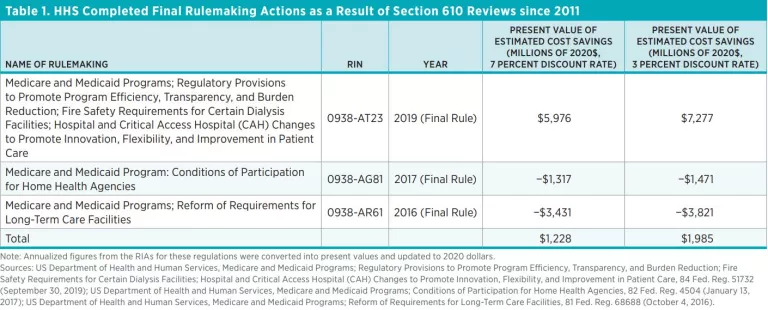
As is evident from table 1, at the time of promulgation two of the regulations were expected to impose net costs (suggesting that it is possible for regulators to impose additional costs on the public as a result of retrospective reviews), but the other regulation was estimated to produce enough savings to more than make up for the net costs imposed by the other two.
On balance, the present value of the net benefits of the three regulations is estimated to be $1.2 billion (in 2020 dollars, at a 7 percent discount rate), according to the economic analyses accompanying these regulations. However, these regulation amended more CFR sections than typical regulation under HHS’s authority. In HHS’s RIA for the final SUNSET rule, HHS notes that one regulation amends five CFR sections on average. However, approximately 130 CFR sections were amended by the three regulations in table 1. Thus, the total benefits and costs reported in table 1 can be thought of as having emanated from 26 average regulations for the purposes of this benefits analysis.
In the RIA for the SUNSET rule, HHS also projects that 53 average regulations are likely to be rescinded and 159 are likely to be amended as a result of the rule. HHS does not provide information about whether savings are more likely to come disproportionately from amended regulations or from rescinded regulations. If one assumes that savings are likely to come from both equally, then these 212 combined updates could be expected to yield $10.0 billion to $16.2 billion in net savings, provided that HHS’s recently completed actions stemming from Section 610 reviews are representative of the benefits likely to follow from the SUNSET rule.
However, any regulations updated in response to the new retrospective review procedure would not be promulgated immediately. Some would likely be finalized in the decade following implementation of the SUNSET rule, and the finalization of some could even extend into the following decade. Assuming that, on average, regulations deliver benefits 10 years in the future, then the present value of these benefits is $5.1 billion (in 2020 dollars) at a 7 percent discount rate and $12.1 billion at a 3 percent discount rate.
Given the uncertainty surrounding this benefit estimate, one could look at other retrospective review efforts in addition to HHS’s Section 610 reviews. Another source of information about the benefits of retrospective review efforts are the regulations promulgated in response to Executive Order 13563. A study of these efforts by the Administrative Conference of the United States identifies three major regulations from HHS that were the result of retrospective review and were included in the 2013 and 2014 Office of Management and Budget reports to Congress on the benefits and costs of federal regulations. Table 2 presents those regulations along with estimates of their impacts, as quantified in their RIAs at the time of promulgation. The preamble of one of the rules (0938-AQ89) notes that the rule’s provisions meet the objectives of Section 610 of the RFA. However, the rule is not labeled as resulting from a Section 610 review in HHS semiannual agendas, which explains why it is left out of table 1.
As a group, at the time of their promulgation, the three rules were expected to achieve net benefits of $4,640 million at a 7 percent discount rate and $5,227 at a 3 percent discount rate (in 2020 dollars). Those rulemakings amend between 120 and 180 CFR sections. Taking the midpoint of this range suggests these rules amended 30 average rulemakings. Assuming that 212 rulemaking updates occur in coming years, these could be expected to yield $32.8 billion to $36.9 billion in benefits. If these benefits arrive in 10 years, then the present value of these benefits is $16.7 billion at a 7 percent discount rate and $27.5 billion at a 3 percent discount rate.
If one assumes that the entire sample of rules in tables 1 and 2 should be considered together, then the combined estimated net benefits of the regulations are $5,868 million to $7,212 million, amending approximately 280 sections of the CFR. Taking into account that an average rulemaking amends five sections, and assuming that benefits arrive 10 years in the future, this approach yields a present value of estimated benefits of $11.3 billion at a 7 percent discount rate and $20.3 billion at a 3 percent discount rate.

Sensitivity
The secretary of Health and Human Services has the option to extend expiration dates, so there is a chance that benefits will be pushed further into the future, which would lower the present value of these benefits. If the review of a regulations concludes that regulations should be amended or rescinded, then HHS has two years from the date that the findings of the review are published in the Federal Register to amend or rescind the regulation. If the secretary determines that completion of the amendment or rescission is not feasible by the established date, he or she can certify this in a statement published in the Federal Register and then extend the completion date by one year at a time for no more than three times. For sensitivity purposes, one might assume therefore that the benefits estimates, which range from $5.1 billion to $27.5 billion, might arrive three years later as a result of the delay provisions available to the secretary. In that case, benefits would range from a low of $4.2 billion to a high of $25.2 billion.
Context
To put these estimates (which may seem large at first glance) in context, one recent study estimates that the cumulative cost of federal regulations in 2012 was $4 trillion. The $4 trillion federal regulatory cost estimate is derived using an endogenous growth model and data on regulatory restrictions across US industries. Regulatory counts, and specifically counts of regulatory requirements or restrictive terminology, are now often used to evaluate regulatory burdens. For example, Canada recently adopted measures of regulatory burdens across agencies using regulatory counts. These kinds of metrics are employed in states as part of regulatory reform efforts and now appear widely in peer-reviewed academic studies.
In 2012, HHS regulations comprised about 5 percent of federal regulatory restrictions. If HHS restrictions impose on average the same burden as restrictions from the federal government as a whole, then HHS regulations imposed costs of $200 billion in 2012, which, in 2020 dollars, is $228 billion. Meanwhile, $10 billion in benefits annualized over 10 years at a 7 percent discount rate constitutes about $1.4 billion per year in savings, which is less than 1 percent of the annual estimated costs of HHS regulations, suggesting that the benefits estimates earlier are plausible and may even be modest compared to the estimated costs of HHS regulations. It is also worth noting that many of the regulations appearing in tables 1 and 2 amend the Medicare program. Because of that program’s size, it is not surprising that regulations amending it could have impacts in the billions of dollars.
Indirect Benefits
The benefits of the SUNSET rule are not purely financial, as coincident risk reductions are likely to extend from the benefits estimated earlier. One recent study estimates that for every $109 million (in 2019 dollars) in costs resulting from a regulation, one can expect one death to occur. Regulatory costs induce mortality because income reductions reduce expenditures on health and safety, thereby increasing risks to life. Put differently, every $109 million in 2019 dollars (or $111 million in 2020 dollars) HHS saves through its retrospective review efforts will yield one expected life saved. These lives saved (or extended) in turn produce additional cost savings not considered in the previous calculations and are thus a cobenefit of the SUNSET rule.
The fact that HHS regulations cost an estimated $228 billion annually suggests that approximately 2,050 additional expected deaths occur annually as a result of the cost of HHS regulations. Although this increase in mortality does not account for how the HHS’s regulations may reduce mortality, it is unlikely that the costs and the benefits of HHS policy all fall on the same individuals, so these effects deserve attention in their own right.
The cost savings in the discussion of benefits earlier can be used to estimate the coincident health benefits—as well as their corresponding cost savings—that serve as additional cobenefits of the SUNSET rule. These cobenefits help to reduce any overall negative health consequences imposed by the costs of HHS’s regulations. For example, the $5.1 billion to $12.1 billion in projected cost savings based on HHS’s past Section 610 reviews yields an indirect benefit of 46 to 109 initial expected lives saved (this range is “initial” because it is a present value). Assuming saved individuals contribute roughly the average amount of goods or services an American produces in their remaining lifespan, extending each life saved yields an additional return (in terms of cost savings) of $1.1 million on average, which cumulatively yields another $50.6 million to $119.9 million in additional cost savings.
The cobenefits will be offset to a modest extent by the costs of the SUNSET rule and could also be offset if there are unquantified risk increases that stem from the rule. However, even at the high end of HHS’s projected total cost estimates, which are around $200 million in present value terms, costs may not induce more than two initial deaths, meaning that the net risk reduction anticipated from the rule may not differ significantly from the gross risk reduction.
Although these cobenefits are relatively minor compared with the direct benefits anticipated from the SUNSET rule, the cobenefits alone may be large enough to exceed the estimated costs of the rule. Furthermore, because HHS intends to assess and (if necessary) review most important health and safety regulations, it is possible that regulations that reduce risks on balance will not be allowed to be rescinded to offset these estimated indirect cobenefits. The estimated indirect cobenefits of the SUNSET rule appear in table 3.

Net Benefits
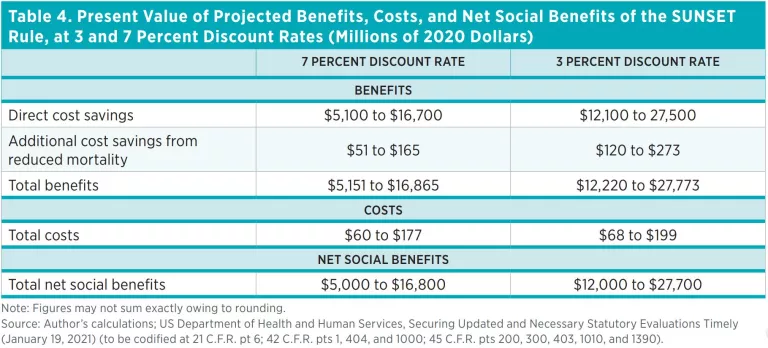
Table 4 aggregates the direct benefits and cobenefits estimated in this analysis with the costs estimated in the SUNSET rule RIA. Total benefits are estimated to range from $5.2 billion to $27.8 billion. On an annualized basis, the benefits are expected to range from $740 million to $3.3 billion annually over a 10-year time horizon, depending on the discount rate used. Meanwhile, total costs range from $60 million to $199 million. The costs are expected to range from $7.9 million to $25.2 million on an annualized basis. Thus, benefits are expected to exceed costs by about two orders of magnitude. The present value of the net benefits expected from the SUNSET rule range from $5.0 billion to $27.7 billion (in 2020 dollars) depending on the discount rate used and the source of the benefit estimate.
Discussion and Uncertainty
The estimates of benefits presented earlier are surrounded by a great deal of uncertainty because it is impossible to know which regulations will be affected by the SUNSET rule. It is, therefore, worth revisiting some of the assumptions underlying the benefits estimates. First, this analysis assumes that past retrospective reviews such as those resulting from past Section 610 reviews or as a result of Executive Order 13563 are likely to be representative of future review efforts under the SUNSET rule. This could turn out not to be true. And while these past reviews also offer some reason to be optimistic that substantial savings are already being achieved even without a new forcing provision, the analysis here also suggests the net benefits of reviews could be increased substantially if the number of reviews were increased. This would likely occur with stronger enforcement mechanisms, such as a sunset provision.
Also, the prospective forecasts of the effects of rules emanating from these past review efforts could turn out not to be correct. For example, sometimes important cost or cost savings estimates are left out of HHS regulatory analyses. Hence, an implicit assumption in the calculations earlier is that the RIAs for those rules were produced competently and absent political interference, which may not be the case. However, uncertainty in prospective analyses is also one of the primary reasons for conducting more retrospective reviews, an aim of the SUNSET rule. Moreover, the net benefits stated earlier are so large that billions of net savings could be wiped out and the net benefits would still be positive. For example, 50 percent of the combined benefits from the regulations identified as cost saving in tables 1 and 2 could be wiped out and cumulatively the projected net benefits of the SUNSET rule would still be over $1 billion.
A plausible way that the SUNSET rule could produce negative net benefits is HHS using the enhanced review process to impose additional regulations with negative net benefits. Sometimes regulators use retrospective review efforts as a justification to move forward with policies that were already a priority for other reasons. To the extent that the rule facilitates such efforts, it could impose net costs. That said, to some extent this issue has already been considered, since some of the rules appearing in tables 1 and 2 were expected at the time of their promulgation to impose net costs. Moreover, to the extent HHS uses retrospective review efforts as a justification to move forward with policies that were already a priority, such regulations may have been likely to be promulgated even absent the SUNSET rule. HHS is likely to impose costly regulations with or without an enhanced retrospective review process, and it seems more likely that HHS will choose to reduce burdens on balance if it has stronger incentives to conduct retrospective review.
Another source of uncertainty relates to HHS’s cost estimates, which, although modest relative to estimated benefits, may actually be overstated in the RIA for the SUNSET rule. The largest cost identified by HHS is the estimated cost of monitoring, which essentially involves the writing of comments and tracking of HHS regulatory activities by interested members of the public. However, HHS has not taken into account the cost of rent-seeking. To the extent that lobbying for anticompetitive regulations is displaced by having to monitor HHS’s new regulatory reviews and write additional comments, this may well constitute a social benefit to society as a whole (even if it constitutes a private cost to the monitors). The analysis in this brief has not attempted to quantify the costs of this rent-seeking but notes that benefits may be underestimated here (or, similarly, costs may be overestimated in the RIA) if rent-seeking activity is reduced by the SUNSET rule.
A final source of uncertainty is the small size of the sample of rules used to project the future benefits of regulations emanating from retrospective review. This small sample size results from there not being a large number of rules updated in response to reviews and from few rules having any economic analysis associated with them, regardless of whether they are the result of retrospective review or any other reason. The sample size certainly leads to questions about the precision of the benefits estimates here. However, a goal of the SUNSET rule is to stimulate retrospective analysis (although perhaps not a complete cost-benefit analysis in most cases). Thus, conceivably a larger sample of regulations will be available for future studies of the benefits and costs of retrospective review owing to the SUNSET rule.
Conclusion
Overall, there is considerable uncertainty with respect to this analysis. In general, the numbers stated earlier should be taken with caution, since HHS is not going to be updating the same regulations in response to future reviews as it did in response to past reviews, and there is considerable uncertainty about whether these past efforts were as successful as their forward-looking analyses projected at the time of their implementation.
That said, the potential for billions of dollars in net benefits is realistic, especially given the reach and burden of HHS rules across the economy. Benefits could conceivably extend into the tens of billions of dollars, dwarfing costs that are in the tens to low hundreds of millions. Given the vast discrepancy between estimated benefits and estimated costs, it is not surprising that HHS has concluded that it is appropriate to move forward with the SUNSET rule. The rule may also lead to substantial health benefits for the public as an indirect consequence of the cost savings that future review efforts are likely to uncover.
Ultimately, the success of the SUNSET rule will depend on the civil servants tasked with executing it. However, a forcing mechanism such as a sunset provision seems likely to ensure that more good-faith efforts at retrospective review actually occur.

